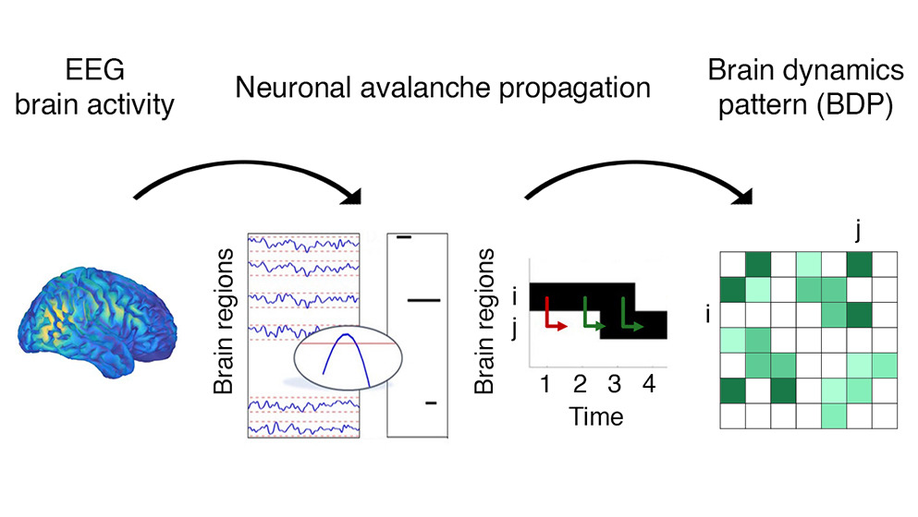
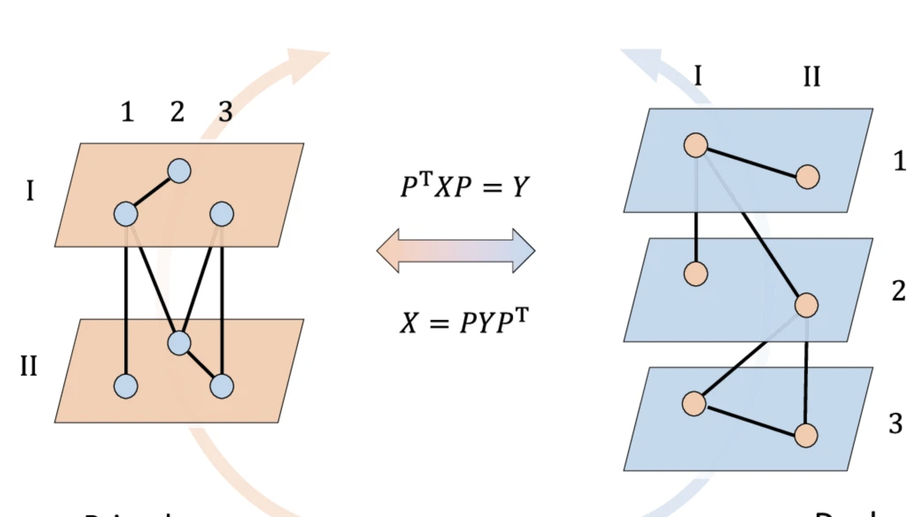
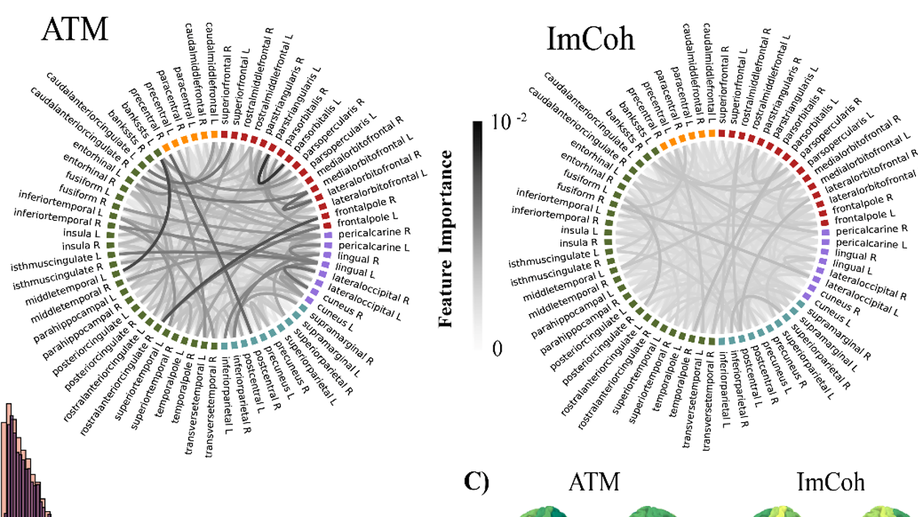
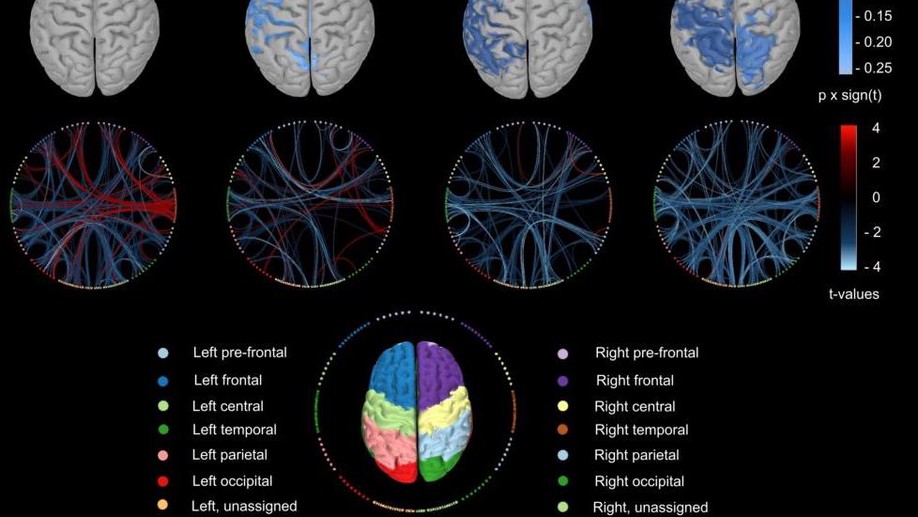
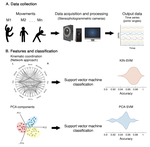
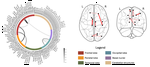
About me
I am an Inria research scientist at Paris Brain Institute in the NERV Lab.
My research currently focuses on the development of tools to address the “Brain-Computer Interface (BCI) inefficiency” issue, reflected by a non-negligible portion of users who cannot control the device even after extensive training. I essentially consider two main approaches: the search for neurophysiological markers of BCI training and the integration of multimodal data to enrich the information provided to the classifier. In 2025, I received the Early Career Award from the International BCI Society.
I previously served as secretary general of the French academic association promoting the advances in BCI, called CORTICO, and as co-chair of the Postdocs and Students Committee of the BCI Society.
You can download my CV in pdf.
Don’t hesitate to contact me if you want any additional information or if you are interested by a research collaboration!
Interests
- Closed-loop systems (Brain-Computer Interfaces & Neurofeedback)
- Linear and non-linear functional connectivity
- Multimodal integration
- Machine learning
- Biomedical instrumentation
Education
PhD in Biomedical instrumentation, 2015
CEA-LETI (Grenoble, France)
MSc in Neuropsychology and Clinical Neurosciences, 2015
Grenoble Alpes University
MEng in Information and Communications Technology for Health, 2012
IMT Atlantique (Brest, France)